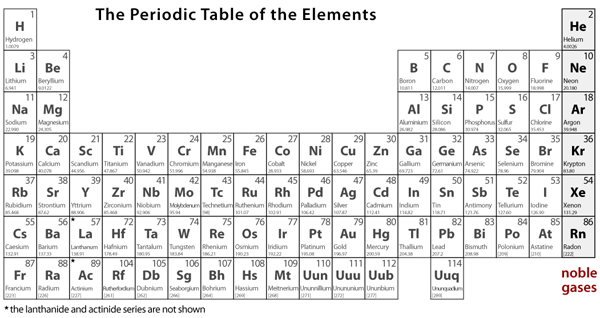
The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. By following the standard notation, you can avoid ambiguity in parsing and obtain accurate results.\). Scientists use the periodic table to quickly refer to information about an element, like atomic mass and chemical symbol. For example, C2H5OH for ethanol, H2O for water, or Na2SO4 for sodium sulfate. As such, it’s highly unstable and unable to exist for very long. It is element 94, making it bigger than uranium. 
In 1940, a new element, plutonium, was created in a laboratory. Element 92 is uranium, the biggest element that occurs naturally on Earth. It is important to ensure that the chemical formula is entered correctly using standard notation. Before the year 1940, it was believed that only 92 elements existed. The result table includes the element symbol, atomic mass, multiplier (indicating the number of atoms of each element in the formula), and the calculated result. In addition to the molar mass, the calculator also provides a result table with detailed information on the individual elements in the formula. The atomic number of each element increases by one, reading from left to right. The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. To use the calculator, simply input the chemical formula of the substance and click the 'Calculate' button. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. PeriodA horizontal row in the periodic table. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) 28.02 amu. Members of a group typically have similar properties and electron configurations in their outer shell. It then calculates the molar mass by adding up the atomic weights of all the elements in the substance. GroupA vertical column in the periodic table. To use the calculator, simply input the chemical formula of the substance and click the "Calculate" button. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. It can supply you with a correct representation of a substance’smass and size, and valence electron shell. 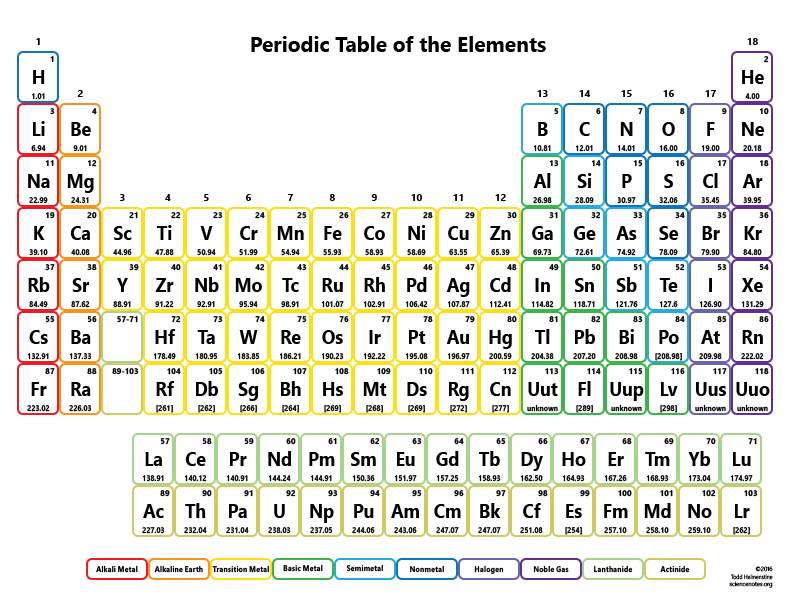
The Molar Mass Calculator is a tool used to calculate the molar mass of a substance based on its chemical formula. Periodic Table With Atomic Or Molar The Routine Kitchen table is an integral part of the study of research, and it may be useful in identifying a substance’s attributes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
